Isomerization
In chemistry, isomerization or isomerisation is the process in which a molecule, polyatomic ion or molecular fragment is transformed into an isomer with a different chemical structure.[1] Enolization is an example of isomerization, as is tautomerization.[2] When the isomerization occurs intramolecularly it may be called a rearrangement reaction.[citation needed]
When the activation energy for the isomerization reaction is sufficiently small, both isomers will exist in a temperature-dependent equilibrium with each other. Many values of the standard free energy difference, , have been calculated, with good agreement between observed and calculated data.[3]
Examples and applications
Alkanes
Skeletal isomerization occurs in the cracking process, used in the petrochemical industry. As well as reducing the average chain length, straight-chain hydrocarbons are converted to branched isomers in the process, as illustrated the following reaction of n-butane to i-butane.[citation needed]
Fuels containing branched hydrocarbons are favored for internal combustion engines for their higher octane rating.[4]
Alkenes
Terminal alkenes isomerize to internal alkenes in the presence of metal catalysts. This process is employed in the Shell higher olefin process to convert alpha-olefins to internal olefins, which are subjected to olefin metathesis. In certain kinds of alkene polymerization reactions, chain walking is an isomerization process that introduces branches into growing polymers.[citation needed]
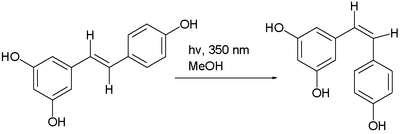
The trans isomer of resveratrol can be converted to the cis isomer in a photochemical reaction.[5]
Thermal rearrangement of azulene to naphthalene has been observed.[citation needed]
Other examples
Aldose-ketose isomerism, aka Lobry de Bruyn–van Ekenstein transformation, provides an example in saccharide chemistry.[citation needed]
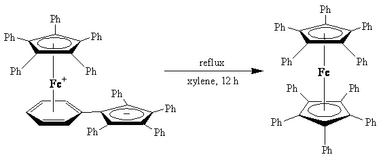
An example of an organometallic isomerization is the production of decaphenylferrocene, [(η5-C5Ph5)2Fe] from its linkage isomer.[6][7]
See also
References
- ^ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "isomerization". doi:10.1351/goldbook.I03295
- ^ Antonov L (2016). Tautomerism: Concepts and Applications in Science and Technology (1st ed.). Weinheim, Germany: Wiley-VCH. ISBN 978-3-527-33995-2.
- ^ How to Compute Isomerization Energies of Organic Molecules with Quantum Chemical Methods Stefan Grimme, Marc Steinmetz, and Martin Korth J. Org. Chem.; 2007; 72(6) pp 2118 - 2126; (Article) doi:10.1021/jo062446p
- ^ "Hydrocarbons". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. 2002. doi:10.1002/14356007.a13_227. ISBN 3527306730.
{{cite encyclopedia}}: Cite uses deprecated parameter|authors=(help) - ^ Resveratrol Photoisomerization: An Integrative Guided-Inquiry Experiment Elyse Bernard, Philip Britz-McKibbin, Nicholas Gernigon Vol. 84 No. 7 July 2007 Journal of Chemical Education 1159.
- ^ Brown, K. N.; Field, L. D.; Lay, P. A.; Lindall, C. M.; Masters, A. F. (1990). "(η5-Pentaphenylcyclopentadienyl){1-(η6-phenyl)-2,3,4,5-tetraphenylcyclopentadienyl}iron(II), [Fe(η5-C5Ph5){(η6-C6H5)C5Ph4}], a linkage isomer of decaphenylferrocene". J. Chem. Soc., Chem. Commun. (5): 408–410. doi:10.1039/C39900000408.
- ^ Field, L. D.; Hambley, T. W.; Humphrey, P. A.; Lindall, C. M.; Gainsford, G. J.; Masters, A. F.; Stpierre, T. G.; Webb, J. (1995). "Decaphenylferrocene". Aust. J. Chem. 48 (4): 851–860. doi:10.1071/CH9950851.