Thyroid peroxidase
| iodide peroxidase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 iodide peroxidase monomer, Zobellia galactanivorans | |||||||||
| Identifiers | |||||||||
| EC no. | 1.11.1.8 | ||||||||
| CAS no. | 9031-28-1 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
| thyroid peroxidase | |||||||
|---|---|---|---|---|---|---|---|
| Identifiers | |||||||
| Symbol | TPO | ||||||
| NCBI gene | 7173 | ||||||
| HGNC | 12015 | ||||||
| OMIM | 606765 | ||||||
| RefSeq | NM_175722 | ||||||
| UniProt | P07202 | ||||||
| Other data | |||||||
| EC number | 1.11.1.8 | ||||||
| Locus | Chr. 2 pter-p24 | ||||||
| |||||||
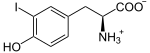
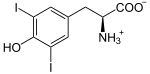
Thyroid peroxidase, also called thyroperoxidase (TPO), thyroid specific peroxidase or iodide peroxidase, is an enzyme expressed mainly in the thyroid where it is secreted into colloid. Thyroid peroxidase oxidizes iodide ions to form iodine atoms for addition onto tyrosine residues on thyroglobulin for the production of thyroxine (T4) or triiodothyronine (T3), the thyroid hormones.[1] In humans, thyroperoxidase is encoded by the TPO gene.[2]
Catalyzed reaction
[edit]Iodide is oxidized to iodine radical which immediately reacts with tyrosine.
The second iodine atom is added in similar manner to the reaction intermediate 3-iodotyrosine.
Function
[edit]
Inorganic iodine enters the body primarily as iodide, I−. After entering the thyroid follicle (or thyroid follicular cell) via a Na+/I− symporter (NIS) on the basolateral side, iodide is shuttled across the apical membrane into the colloid via pendrin, after which thyroid peroxidase oxidizes iodide to atomic iodine (I) or iodinium (I+). The "organification of iodine," the incorporation of iodine into thyroglobulin for the production of thyroid hormone, is nonspecific; that is, there is no TPO-bound intermediate, but iodination occurs via reactive iodine species released from TPO.[4] The chemical reactions catalyzed by thyroid peroxidase occur on the outer apical membrane surface and are mediated by hydrogen peroxide.
Stimulation and inhibition
[edit]TPO is stimulated by TSH, which upregulates gene expression.
TPO is inhibited by the thioamide drugs, such as propylthiouracil and methimazole.[5] In laboratory rats with insufficient iodine intake, genistein has demonstrated inhibition of TPO.[6]
Clinical significance
[edit]Thyroid peroxidase is a frequent epitope of autoantibodies in autoimmune thyroid disease, with such antibodies being called anti-thyroid peroxidase antibodies (anti-TPO antibodies). This is most commonly associated with Hashimoto's thyroiditis. Thus, an antibody titer can be used to assess disease activity in patients that have developed such antibodies.[7][8]
Diagnostic use
[edit]In diagnostic immunohistochemistry, the expression of thyroid peroxidase (TPO) is lost in papillary thyroid carcinoma.[9]
References
[edit]- ^ Ruf J, Carayon P (Jan 2006). "Structural and functional aspects of thyroid peroxidase". Archives of Biochemistry and Biophysics. 445 (2): 269–77. doi:10.1016/j.abb.2005.06.023. PMID 16098474.
- ^ Kimura S, Kotani T, McBride OW, Umeki K, Hirai K, Nakayama T, Ohtaki S (Aug 1987). "Human thyroid peroxidase: complete cDNA and protein sequence, chromosome mapping, and identification of two alternately spliced mRNAs". Proceedings of the National Academy of Sciences of the United States of America. 84 (16): 5555–9. Bibcode:1987PNAS...84.5555K. doi:10.1073/pnas.84.16.5555. PMC 298901. PMID 3475693.
- ^ Walter F., PhD. Boron (2003). Medical Physiology: A Cellular And Molecular Approaoch. Elsevier/Saunders. p. 1300. ISBN 1-4160-2328-3.
- ^ Kessler J, Obinger C, Eales G (Jul 2008). "Factors influencing the study of peroxidase-generated iodine species and implications for thyroglobulin synthesis". Thyroid. 18 (7): 769–74. doi:10.1089/thy.2007.0310. PMID 18631006.
- ^ Nagasaka A, Hidaka H (Jul 1976). "Effect of antithyroid agents 6-propyl-2-thiouracil and 1-methyl-2-mercaptoimidazole on human thyroid iodine peroxidase". The Journal of Clinical Endocrinology and Metabolism. 43 (1): 152–8. doi:10.1210/jcem-43-1-152. PMID 947933.
- ^ Doerge DR, Sheehan DM (Jun 2002). "Goitrogenic and estrogenic activity of soy isoflavones". Environmental Health Perspectives. 110 (Suppl 3): 349–53. doi:10.1289/ehp.02110s3349. PMC 1241182. PMID 12060828.
- ^ McLachlan SM, Rapoport B (2000). "Autoimmune response to the thyroid in humans: thyroid peroxidase--the common autoantigenic denominator". International Reviews of Immunology. 19 (6): 587–618. doi:10.3109/08830180009088514. PMID 11129117. S2CID 11431166.
- ^ Chardès T, Chapal N, Bresson D, Bès C, Giudicelli V, Lefranc MP, Péraldi-Roux S (Jun 2002). "The human anti-thyroid peroxidase autoantibody repertoire in Graves' and Hashimoto's autoimmune thyroid diseases". Immunogenetics. 54 (3): 141–57. doi:10.1007/s00251-002-0453-9. PMID 12073143. S2CID 2701974.
- ^ Tanaka T, Umeki K, Yamamoto I, Sugiyama S, Noguchi S, Ohtaki S (May 1996). "Immunohistochemical loss of thyroid peroxidase in papillary thyroid carcinoma: strong suppression of peroxidase gene expression". The Journal of Pathology. 179 (1): 89–94. doi:10.1002/(SICI)1096-9896(199605)179:1<89::AID-PATH546>3.0.CO;2-R. PMID 8691351. S2CID 26045198.
External links
[edit]- Thyroid+Peroxidase at the U.S. National Library of Medicine Medical Subject Headings (MeSH)