Thiazolidine

| |
| Names | |
|---|---|
| Preferred IUPAC name
1,3-Thiazolidine[1] | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.007.275 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C3H7NS | |
| Molar mass | 89.16 g·mol−1 |
| Density | 1.131 g/cm3[2] |
| Boiling point | 72 to 75 °C (162 to 167 °F; 345 to 348 K)[2] at 25 torr |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Thiazolidine is a heterocyclic organic compound with the formula (CH2)3(NH)S. It is a 5-membered saturated ring with a thioether group and an amine group in the 1 and 3 positions. It is a sulfur analog of oxazolidine. Thiazolidine is a colorless liquid. Although the parent thiazolidine is only of academic interest, some derivatives, i.e., the thiazolidines, are important, such as the antibiotic penicillin.
Preparation
[edit]Thiazolidine is prepared by the condensation of cysteamine and formaldehyde.[3] Other thiazolidines may be synthesized by similar condensations. A notable derivative is 4-carboxythiazolidine (thioproline), derived from formaldehyde and cysteine.
Derivatives
[edit]
Useful thiazolidines include the drug pioglitazone, the antibiotic penicillin, and N-Methyl-2-thiazolidinethione, an accelerator for the vulcanization of chloroprene rubbers.[4]
Thiazolidines functionalized with carbonyls at the 2 and 4 positions, the thiazolidinediones, are drugs used in the treatment of diabetes mellitus type 2. Rhodanine is a related bioactive species, featuring one carbonyl and one thiocarbonyl.
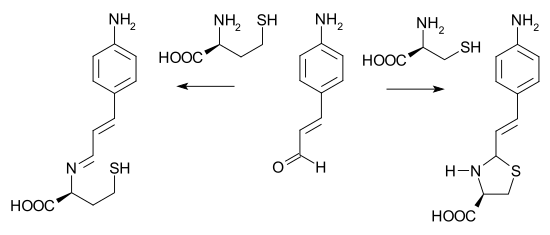
Many thiazolidines are prepared from cysteine and related aminothiols.[5] p-aminocinnamaldehyde is able to differentiate between cysteine and homocysteine. With cysteine, a buffered water solution of the aldehyde changes from yellow to colorless due to a secondary ring closing reaction of the imine. Homocysteine is unable to give ring closure and the color does not change.
See also
[edit]References
[edit]- ^ International Union of Pure and Applied Chemistry (2014). Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013. The Royal Society of Chemistry. p. 142. doi:10.1039/9781849733069. ISBN 978-0-85404-182-4.
- ^ a b Thiazolindine
- ^ Ratner, Sarah; Clarke, H. T. (1937). "Action of formaldehyde on cysteine". Journal of the American Chemical Society. 59: 200–6. doi:10.1021/ja01280a050.
- ^ Rüdiger Schubart (2000). "Dithiocarbamic Acid and Derivatives". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a09_001. ISBN 3527306730.
- ^ Wang, Weihua; Rusin, Oleksandr; Xu, Xiangyang; Kim, Kyu Kwang; Escobedo, Jorge O.; Fakayode, Sayo O.; Fletcher, Kristin A.; Lowry, Mark; Schowalter, Corin M.; Lawrence, Candace M.; Fronczek, Frank R.; Warner, Isiah M.; Strongin, Robert M. (2005). "Detection of Homocysteine and Cysteine". Journal of the American Chemical Society. 127 (45): 15949–15958. doi:10.1021/ja054962n. PMC 3386615. PMID 16277539.